Strategizing from 7 cities across the globe
Optimising Neonatal Abstinence Syndrome Care from Clinical Admission to Discharge
Wael Abdul Khalek
Edited by: Uma Mei Terpend & Selina Riachi


Introduction
Neonatal Abstinence Syndrome (NAS) is a baby’s withdrawal symptoms when exposed to opioids and other substances such as benzodiazepines, nicotine, alcohol, or selective serotonin reuptake inhibitors (SSRIs). The non-opioid substances also worsen the symptoms when paired with opioids during pregnancy. Affected infants exhibit symptoms such as hyperirritability, high pitched crying, jitteriness, tremor and disturbed sleep. Additionally, the baby may experience symptoms in the gastrointestinal (GI) tract (as typically seen in heroin withdrawal) such as regurgitation, vomiting, poor feeding, and diarrhea (Anbalagan et al., 2024a). Countries like the United States (US) are experiencing an opioid crisis due to illicit drug use and prescription (Ayoo et al., 2020); and as a result, the number of NAS babies has surged over the years, causing many implications. First of all, a 17-day stay in the hospital due to NAS can cost around $66,700 (Writers, 2018), placing a major burden on parents. In addition, NAS has been correlated to different neurodevelopmental disorders such as autism. It is imperative to emphasize that NAS is not exclusive to neonates birthed from mothers with opioid use disorder. Some pregnant women may be prescribed medications crucial for their wellbeing, such as antidepressants which manage and prevent a relapse of depression during pregnancy and post-partum, which shows us that newborns may be exposed to these substances in utero for different reasons outside of addiction. Thus, it is important to support both the mother and the child through certain evidence-based treatment plans, since the mothers that are exposed to these substances may be experiencing trauma, recovery, mental health challenges, or socioeconomic instability. There are two assessment methods of interest which significantly differ in approach: the Finnegan Neonatal Abstinence Scoring System (FNASS), and the Eat Sleep Console method (ESC) (Gomez-Pomar & Finnegan, 2018).
The FNASS tests the severity of 21 symptoms in the neonate and creates a score that guides pharmacological guidance (pharmacological intervention initiated if scores reach ≥ 12 or if three scores of ≥ 8 are consecutively recorded (Gomez-Pomar & Finnegan, 2018)). This often results in the administration of morphine or other medications with the same effect. The FNASS usually gets the job done, yet it still remains somewhat inefficient since the symptoms may be subjectively interpreted, and it is specifically designed only for opioids (NAS may be caused and complicated by different substances such as benzodiazepines, SSRIs and SNRIs…) (Neonatal Abstinence Syndrome: MedlinePlus Medical Encyclopedia, n.d.). In contrast, the ESC method emphasizes the baby’s ability to eat, sleep, and be consoled. Despite its simplicity, the current studies on ESC have shown that there has been a significant decrease in hospital stays and opioid exposure for NAS babies when this method is used, making it an effective alternative to FNASS which doesn’t overemphasize the need for pharmacologic intervention when not necessary. However, ESC is heavily dependent on caregiver availability, may mask some underlying withdrawal symptoms, and may not be reliable with specific types of neonates, such as in cases where preterm infants or infants are exposed to fentanyl in utero (Wharton et al., 2021).
Thus, the current options for NAS treatment rely on models that either over-medicalize withdrawal or fail to shed light on underlying neurobiological distress. Accordingly, the goal of this paper is to build a process-based treatment framework for NAS that weaves together neurobiological understanding and substance heterogeneity, staffing and maternal availability, and socioeconomic restraints throughout the full hospitalization process (admission to discharge).
To accomplish this goal, this study examines the existing limitations of current NAS assessment methods across different clinical and sociocultural contexts, defines clinical outcome targets, and presents a full structured framework of the new treatment method aligned with the hospital workflow, followed by a discussion of limitations and future validation.
I. Implications of Pathophysiology of NAS on Treatment Approach
The main mechanism behind NAS involves transplacental drug transfer, causing the fetus to be physiologically dependent on the drug upon the abrupt cessation of the drug transfer at birth (Anbalagan et al., 2024a). In terms of etiology, opioids such as heroin, morphine, and prescription opioids are the most frequently identified agents. In addition, there are some synthetic and semi-synthetic opioids such as methadone and buprenorphine that are intentionally prescribed during pregnancy and are used to treat opioid use disorder (medication-assisted therapy) (Anbalagan et al., 2024a). These opioids are meant to prevent relapse and withdrawal in the mother but may possibly also trigger the fetus to be dependent. However, untreated opioid use by the mother is a far greater risk than MAT, which is shown through studies that compared the likelihood of NAS between methadone-exposed neonates and neonates that are exposed to synthetic and semi-synthetic opioids. The studies demonstrate that methadone-exposed infants were 6 times more likely to be pharmacologically treated than MAT-exposed infants (Anbalagan et al., 2024a). When an infant is chronically exposed to opioids, the cAMP (cyclic adenosine monophosphate) pathways are upregulated and receptors in the locus coeruleus are also stimulated to counteract the effect of the opioid (Anbalagan et al., 2024a). After cessation of drug transfer, many neurotransmitter levels are alienated: norepinephrine levels significantly increase, which causes most of the withdrawal symptoms; serotonin and dopamine levels also drop, causing sleep disturbances and increased irritability; and an increase in acetylcholine is linked to symptoms like diarrhea and sneezing. All these neurotransmitter imbalances may cause a wide array of symptoms such as high-pitched crying, tremors, irritability, sleep fragmentation, feeding difficulties, and temperature instability (Anbalagan et al., 2024a). Exposure to other substances in utero may have a similar effect, but there are distinctions between the mechanisms of each substance.
From a physiological perspective, both FNASS and ESC have their merits and weaknesses when it comes to assessing and treatment of NAS. To start, the FNASS is well structured and categorized, assessing the effect of the syndrome on the CNS, GI tract, and metabolic, vasomotor, and respiratory systems, by linking them to the previously mentioned systems. Then, a score is formed consistently to quantify the effect of the withdrawal on the baby’s physiology and how administration of a certain drug affects withdrawal (NCPOEP, 2018). However, due to the vast range of symptoms produced by NAS, the score can rise due to mild symptoms, reaching the threshold that calls for pharmacologic treatment; in this case, the finnegan scoring may lead to early or over-intervention by quantifying these mild symptoms. In addition, the nature of the assessment tool in and of itself may lead to poor reliability due to the subjectivity in the way the symptoms are perceived (Rech et al., 2024) This may reflect a mismatch between the score produced from observation and the pathophysiology of NAS because the latter causes continuous autonomic dysregulation (since opioids were calming the nervous system prior to labour), meaning that the symptoms are constantly fluctuating and the FNASS only produces a score of the symptoms every few hours, reducing reliability.
Conversely, in terms of pathophysiology, ESC remedies many of the issues that FNASS faces, as it places a much greater emphasis on non-pharmacologic intervention (Dembeck, 2025). First of all, it only examines the most vital of functions (ability to eat sleep and be consoled) (Dembeck, 2025), leaving little room for subjectivity and minimizing unnecessary pharmacologic intervention while also signalling when the neonatal neuroadaptation is failing and intervention is necessary. In addition, the techniques used to counteract the withdrawal symptoms are proven to have a significant effect on the baby’s physiology. The first technique used is having a low stimulus environment for the baby, which reduces the sensory overload and hyperarousal (caused by norepinephrine surge), and promotes healthier sleep cycles (Dembeck, 2025). Additionally, skin-to-skin contact causes a significant release of oxytocin, which inhibits the release of corticotropin releasing hormone (CRH) from the paraventricular nucleus of the hypothalamus (Handlin et al., 2009). Oxytocin also travels to the anterior pituitary gland, inhibiting the release of adrenocorticotropic hormone (ACTH) which reduces the production of cortisol. Cortisol and CRH are both major triggers for the HPA axis stress responses which are significantly active during withdrawal, making frequent release of oxytocin useful in reducing stress and withdrawal symptoms in the neonate (Handlin et al., 2009). Thus, it can be concluded that the ESC method is efficient in terms of reducing hyperarousal and hyperirritability, and its simple criteria leaves little room for interpretation by different healthcare workers. However, the physiologic distress that is revealed from the 21 symptoms should also play a role in guiding pharmacologic and nonpharmacologic treatment.
II. Caregiver Availability Factors Affecting NAS Care
The presence of a parental figure plays a crucial role in the outcomes of NAS. In fact, studies have shown a negative correlation between parental presence (mostly maternal) and the need for pharmacologic intervention (Amin et al., 2023). The ESC approach, which is primarily a family cantered approach, encourages caregivers to console their babies. This approach has been proven to be effective in multiple different areas, but ESC’s outcomes are also significantly affected by the implementation conditions. A study conducted at a regional referral centre examined NAS management before and after ESC intervention, focusing on pharmacologic and non-pharmacologic care, caregiver presence, and length of stay (LOS) (Amin et al., 2023). In figure 1, it is shown that the difference between the overall pharmacologic treatment and the LOS before ESC (FNASS) and after ESC assessments and interventions are statistically insignificant. The authors linked this to structural barriers, limited rooming in space in the hospital, and geographic distance, which result in the limited parental presence which is essential to ESC (Amin et al., 2023). This indicates the possibility that caregiver-dependent strategies like ESC may not provide consistent advantages across different healthcare settings and populations. Reliance on caregiver engagement as a key mechanism of care may unintentionally reduce treatment efficacy and efficiency when parental availability is limited by institutional, regional, and socioeconomic factors. This emphasizes the need for maintenance of family-centred care while continuing to be successful in situations when it's not possible to assume constant parental presence. This is especially crucial since parental substance exposure happens in very delicate and complex social backgrounds, such as parental substance use and mental health challenges and addiction recovery, and unstable housing, which all may adversely affect parental presence.


Download the full document

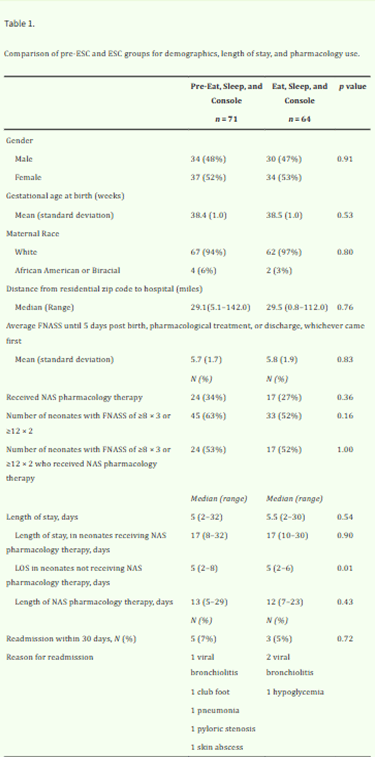
Figure 1: Table comparing pre-ESC and ESC groups for demographics, LOS, and pharmacology use
III. Socioeconomic factors affecting NAS Care
Housing stability, status of employment, level of education, and poverty/class level are all socioeconomic factors that influence the care outcomes of NAS and the efficacy of these tools in different countries.
a) Housing stability
Studies in the United States have shown that mothers of NAS babies usually reside in the lowest SES (socioeconomic status) neighborhoods or don’t have a fixed address, which usually signifies homelessness (Lisonkova et al., 2024). Additionally, it is reported that 30% of discharged NAS neonates were placed into foster/adoptive care (Lisonkova et al., 2024). Infant outcomes are often improved when there is maintenance of a stable home and access to the necessary resources of care and support (Acosta, 2022). However, that is not necessarily the case with these infants, so it is important to ensure housing stability through family housing services or residential transfer programs since it reduces stressors on parents and possibly reduces relapse or substance abuse prior to birth (Dumbhare & Taksande, 2023). On the other hand, a study in Shiraz has shown that 40% of NAS infants (usually of tenants or homeless mothers) were discharged to social services; these infants had longer hospital stays and lower finnegan scores (low NAS severity). This reveals that the housing insecurity that many of these families face actually delayed discharge and increased separation between the parents and infants for legal and custody reasons rather than medical needs.
b) Employment Status
In the US, counties with the highest unemployment rates were strongly correlated with the incidence of NAS (a 10-year high unemployment rate had an incidence ratio of 1:1.11) (Patrick, 2019). However the opposite may also be true: while unemployed mothers may face financial obstacles (transport and care for other children), employed mothers may struggle to find sufficient maternity leave to care for their infant, which may restrict rooming in and different NAS care methods. In European countries, these struggles are buffered by universal healthcare, which is not present in the US, and social welfare systems. Furthermore, European countries provide paid maternity leaves and social support that can help mothers become further involved in care for the baby (Rees et al., 2021). However, this may not always be the case; for example, a study in Scotland noted that parental drug use is associated with job loss (Chandler, 2022), yet the situation stays more manageable due to a greater access to resources.
IV. Structural framework of NAS treatment
a) Admission Phase
Upon admission, staff must verify the detailed maternal history of substance abuse, including the types of substances consumed, timing, and medication history (toxicology data is obtained to identify exposures). Anticipatory care plans such as MAT (providing opioids such as methadone or buprenorphine) are provided to the pregnant woman to minimize harm/miscarriage to the fetus, and stabilise the mother (Anbalagan et al., 2024a). At birth, the pediatric health care provider should monitor the neonate, and after all screenings and tests (ex: apgar scores, head circumference, umbilical cord toxicology...) are performed to ensure stability, the baby is immediately placed in a low stimulus room to combat excitation of CNS at cessation (MacMillan et al., 2018). The baby is then exposed to the first line of treatment: the non-pharmacologic care. This is where the baby is exposed to techniques, such as swaddling and skin-to-skin contact, usually after the baby is admitted to a postpartum. Additionally, the provider should constantly resuscitate the neonate and avoid using opioid antagonists such as naloxone for respiratory depression, as it may worsen NAS symptoms (Canadian Paediatric Society, 2025). Instead, the provider may try to provide supportive respiratory care, such as properly positioning the infant to keep the airway open, supplementing oxygen, performing non-invasive or mechanical ventilation (depending on severity), etc. (Senepete, 2024). Lactation support is also recommended, especially if the mother is under MAT, since it acts as combination of both controlled pharmacologic care and non-pharmacologic care: methadone and/or buprenorphine in milk reduce withdrawal symptoms and the skin-to-skin contact and nutrients facilitate the baby’s development (Haller, 2015). Additionally, a social work consult should be obtained for every NAS case. This consultation sheds light on socioeconomic factors that may worsen NAS symptoms, such as: housing stability, employment status, food securities or other social determinants of health (SDOH). The families in need may then be connected to housing programs or job placement support (Kocherlakota, 2014).
b) Assessment and monitoring
Because FNASS and ESC both have their advantages, both should be used to assess the severity of NAS and to determine the standard for pharmacologic and non-pharmacologic use. Firstly, FNASS should be used to perform an initial scoring of CNS and GI symptoms using electronic medical records (Rech et al., 2024), and maternal availability should be confirmed for rooming-in by educating the mother on the importance of her, or possibly the father’s, participation in the care of the baby (calming CNS, regulating HPA axis and cortisol secretion...) (Handlin et al., 2009). If maternal availability is not present, volunteer social workers can be recruited to aid in tasks such as rooming in, swaddling, and skin to skin contact (Thielking, 2017), but breastfeeding is exclusive to the mother. FNASS should be used for tracking the severity of NAS since it has a detailed array of symptoms that can be used to understand the way the syndrome is developing (delayed symptoms, rapid progression...) (Anbalagan et al., 2024a). However, ESC should be used as the standard for whether pharmacologic treatment such as methadone should be used. At that point, the neonate should be transferred to NICU, especially if they are too unstable (most possible with polysubstance abuse or fentanyl abuse by the mother) (Canadian Paediatric Society, 2025a).
c) Discharge
There are extensive criteria that should be considered for discharge. First of all, the baby should be free of withdrawal symptoms for more than 3-7 days or longer depending on the severity of the opioid used by the mother (MaterNews, n.d.). Additionally, the baby should be properly fed and must show weight gain (one of ESC requirements), and the baby’s vital signs and thermoregulation should be stable throughout the entire recovery period. The AAP and CDC also emphasize that every NAS baby should receive a plan of safe care that assesses family needs and links them to resources, addresses maternal substance treatment, infant care, and home safety (CDC, 2025). Moreover, parents must be educated by the providers on how to take care of the infant to avoid readmission into the hospital. One of the important things that the parents should know is that NAS babies are more likely to experience Sudden Infant Death Syndrome (SIDS). To decrease the possibility of something like SIDS occurring, the parents should be informed that the baby must be placed on his/her back, use a mattress without pillows or loose beddings, and avoid bed-sharing (NCPOEP, 2018).
Conclusion
Neonatal Abstinence Syndrome illustrates a flaw that is present in the current healthcare system: the limited investment in optimizing care plans for infants experiencing NAS. Although new approaches such as ESC have improved NAS care and management, this treatment is hindered by the newborn’s physiological distress, the need for consistent caregiver presence, and socioeconomic challenges for the caregivers. Thus, NAS should not be viewed simply as a condition that affects the newborn, but it should be acknowledged as a condition that is shaped by clinical practice, family availability, and social support systems.
It is also evident that there is no thorough standardized model of care for this syndrome. As such, the goal of this research was to examine these flaws and propose ways to treat NAS while optimizing the care of the patient. The first step that was taken to achieve this was to recognize the advantages and shortcomings of the assessment tools used: the evidence indicates that pharmacologic care should not be entirely guided by FNASS, but the standard should be more based off of the basic functionality of the neonate (ESC). However, ESC was also shown not to be reliable entirely on its own due to its family centered nature and possible underestimation of underlying withdrawal symptoms, making it clear that a hybrid approach to assessment must be used.
Additionally, parent education is very crucial for NAS, since it increases the chance that either the mother or the father participates in the care for the infant, helps avoid future complications at home, and strengthens the family bond/dyad, in turn reducing NAS severity. This also indicates that discharge is not the end of the treatment, and long-term success is purely dependent on parent education, housing support, and access to healthcare.
Future studies can target the other substances that have been correlated with NAS but still to this day have no clear data on their effect on the neonate or whether there is a direct causation between these substances and NAS severity. This is especially relevant since opioid withdrawal has been extensively researched but the effects of substances like fentanyl on neonates and NAS are still not entirely understood (Burduli & Jones, 2025). Additionally, the long term psychosocial and pathophysiological effects of NAS may be further investigated considering there is still no clear data on the physical and mental health of individuals who experienced NAS (Williams, 2023).
References
Anbalagan, S., Falkowitz, D. M., & Mendez, M. D. (2024a). Neonatal abstinence syndrome. StatPearls.
Ayoo, K., Mikhaeil, J. S., Huang, A., & Wąsowicz, M. (2020). The opioid crisis in North America: Facts and future lessons for Europe. Anaesthesiology Intensive Therapy, 52(2), 139–147. https://doi.org/10.5114/ait.2020.94756
Writers, N. (2018, February 27). WV medicaid covers an innovative and less costly treatment model for opioid-affected infants. NASHP. https://nashp.org/wv-medicaid-covers-an-innovative-and-less-costly-treatment-model-for-opioid-affected-infants/
Gomez-Pomar, E., & Finnegan, L. P. (2018). The epidemic of neonatal abstinence syndrome, historical references of its’ origins, assessment, and management. Frontiers in Pediatrics, 6, 33. https://doi.org/10.3389/fped.2018.00033
Wharton, R. E., Casbohm, J. S., Hoffmaster, R., & Johnson, R. C. (2021, January 4). Detection of 30 fentanyl analogs by commercial immunoassay kits. Oxford University Press. https://www.researchgate.net/publication/348215939_Detection_of_30_Fentanyl_Analogs_by_Commercial_Immunoassay_Kits
Neonatal abstinence syndrome (NAS) – NCPOEP. (2018). North Carolina Pregnancy & Opioid Exposure Project. https://ncpoep.org/guidance-document/neonatal-abstinence-syndrome-overview/neonatal-abstinence-syndrome-nas/
Rech, T., Rubarth, K., Bührer, C., Balzer, F., & Dame, C. (2024). The finnegan score for neonatal opioid withdrawal revisited with routine electronic data: Retrospective study. JMIR Pediatrics and Parenting, 7, e50575–e50575. https://doi.org/10.2196/50575
Dembeck, L. (2025, March 18). Eat, sleep, console: Reducing NICU admissions of infants with prenatal opioid exposure – pediatrics nationwide. Pediatrics Nationwide. https://pediatricsnationwide.org/2025/03/18/eat-sleep-console-reducing-nicu-admissions-of-infants-with-prenatal-opioid-exposure/
Handlin, L., Jonas, W., Petersson, M., Ejdebäck, M., Ransjö-Arvidson, A.-B., Nissen, E., & Uvnäs-Moberg, K. (2009). Effects of sucking and skin-to-skin contact on maternal ACTH and cortisol levels during the second day postpartum—influence of epidural analgesia and oxytocin in the perinatal period. Breastfeeding Medicine, 4(4), 207–220. https://doi.org/10.1089/bfm.2009.0001
Amin, A., Frazie, M., Thompson, S., & Patel, A. (2023). Assessing the Eat, Sleep, Console model for neonatal abstinence syndrome management at a regional referral center. Journal of Perinatology : Official Journal of the California Perinatal Association, 43(7), 916–922. https://doi.org/10.1038/s41372-023-01666-9
Lisonkova, S., Wen, Q., Richter, L. L., Ting, J. Y., Lyons, J., Mitchell-Foster, S., Oviedo-Joekes, E., Muraca, G. M., Bayrampour, H., Cattoni, E., & Abrahams, R. (2024). Neonatal abstinence syndrome and infant mortality and morbidity: A population-based study. Frontiers in Pediatrics, 12. https://doi.org/10.3389/fped.2024.1394682
Acosta, S. (2022, February 15). Stable housing is foundational to children’s well-being. Center on Budget and Policy Priorities. https://www.cbpp.org/blog/stable-housing-is-foundational-to-childrens-well-being
Dumbhare, O., & Taksande, A. (2023). Neonatal abstinence syndrome: An insight over impact of maternal substance use. Cureus, 15(10), e47980. https://doi.org/10.7759/cureus.47980
Patrick, M., MPH, MS Stephen W. (2019). Association among county-level economic factors, clinician supply, metropolitan or rural location, and. JAMA, 321(4), 385–393. https://doi.org/10.1001/jama.2018.20851
Rees, P., Carter, B., Gale, C., Petrou, S., Botting, B., & Sutcliffe, A. G. (2021). Cost of neonatal abstinence syndrome: An economic analysis of English national data held in the National Neonatal Research Database. Archives of Disease in Childhood - Fetal and Neonatal Edition, 106(5), 494–500. https://doi.org/10.1136/archdischild-2020-319213
Neonatal abstinence syndrome: MedlinePlus Medical Encyclopedia. (n.d.). https://medlineplus.gov/ency/article/007313.htm
Chandler, A. (2022, May 25). Neonatal abstinence syndrome: Uncertain diagnosis and parent/patient involvement in care. Relations Study | Governing Parental Opioid Use: A Relational Ethnography. https://relations.stir.ac.uk/other-resources/relations-blog/neonatal-abstinence-syndrome-uncertain-diagnosis-and-parent-patient-involvement-in-care/
MacMillan, K. D. L., Rendon, C. P., Verma, K., Riblet, N., Washer, D. B., & Volpe Holmes, A. (2018). Association of rooming-in with outcomes for neonatal abstinence syndrome: A systematic review and meta-analysis. JAMA Pediatrics, 172(4), 345–351. https://doi.org/10.1001/jamapediatrics.2017.5195
Canadian Paediatric Society. (2025a, January 7). Managing newborns at risk for neonatal abstinence syndrome (NAS)/neonatal opioid withdrawal syndrome (NOWS): Updates and emerging best practices. Canadian Paediatric Society. https://cps.ca/en/documents/position/nas-nows
Senepete, J. (2024, July 24). Respiratory distress in preterm infants with nurses for newborns. Nursing Evolutions. https://www.nursingevolutions.com/nurses-for-newborns-managing-respiratory-distress-in-preterm-infants/
Haller, C. (2015). Neonatal Abstinence Syndrome. National Association of State Alcohol and Drug Agency Directors.
Kocherlakota, P. (2014). Neonatal abstinence syndrome. Pediatrics, 134(2), e547–e561. https://doi.org/10.1542/peds.2013-3524
Thielking, M. (2017, January 6). Call in the cuddlers: Volunteers soothe opioid-dependent babies. STAT. https://www.statnews.com/2017/01/06/volunteer-cuddlers-opioids/
Neonatal Abstinence Syndrome. (n.d.). MaterNews. Retrieved January 13, 2026, from https://www.mater.org.au/health-services/our-services/neonatal-abstinence-syndrome
CDC. (2025, November 25). Treat and manage infants affected by prenatal opioid exposure. Opioid Use During Pregnancy.
https://www.cdc.gov/opioid-use-during-pregnancy/treatment/infants-opioid.html
Burduli, E., & Jones, Hendrée, E. (2025). Beyond a simple cause and effect relationship: Exploring the long-term outcomes of children prenatally exposed to opioids and other substances. Seminars in Perinatology, 49(1). https://doi.org/10.1016/j.semperi.2024.152010
Williams, F. (2023, June 20). The long-term effects of neonatal abstinence syndrome. Medical News Today. https://www.medicalnewstoday.com/articles/long-term-effects-of-neonatal-abstinence-syndrome
