Strategizing from 7 cities across the globe
Combatting the Limitations of Chemotherapy: The Importance of Lipid and Gold Nanoparticles in Lung and Brain Cancer Therapies
Nancy Chaya & Yara Haidar
Edited by: Reem Daou


Introduction
Cancer is a life-threatening disease and a major global health challenge, accounting for around 10 million deaths in 2020 (World Health Organization: WHO, 2026). Chemotherapy, one of the most well-known and used treatments for cancer, has severe systemic side effects on patients. To start with, this therapy targets cell division, which is a fundamental cellular process in both healthy and cancerous cells. Consequently, this results in the destruction of both healthy and tumor cells. Even more, chemotherapy is toxic; it can lead to multi-organ failure and numerous visible side effects, including skin rashes, anemia, and nausea. It also can contribute to the development of highly resistant tumors that require different drugs and treatments to control them. Despite these side effects, chemotherapy is still widely used because it is one of the main therapies capable of systemically treating cancer and shrinking tumors rapidly.
However, these limitations necessitate moving towards more targeted, less toxic treatments. One such treatment is nanoparticle-assisted drug delivery. Nanoparticles can be engineered to selectively target tumor cells and ensure high treatment efficacy. Their small size, ranging from 1–100 nanometers, allows them to have a high surface-to-volume ratio and infiltrate cancer cells (Hazarika et al., 2024). Their surfaces can also be modified or coated with specific materials to ensure tissue and cell specificity (Misra et al., 2010).
Our research aims to explore the following question: How can lipid (such as solid lipid nanoparticles and nanostructured lipid carriers) and gold nanoparticles (including nanospheres, nanocages, and nanorods) achieve targeted drug delivery in cancer therapy while minimizing damage to healthy cells? Through this, we hope to showcase the potential and importance of nanoparticles in cancer treatment while providing clear explanations about their mode of action and effectiveness. However, due to the large diversity of cancer types, we will mainly focus on the effects of these two nanoparticles on brain and lung tumors. These cancers are challenging to treat because they involve organs that are central to sustaining life, thus ensuring lower toxicity levels is critical. Moreover, the presence of the blood-brain barrier (BBB) limits the ability of chemotherapy to penetrate brain tissue and necessitates the use of targeted drug delivery systems.
Lipid Nanoparticles
Among the various nanotechnology-based drug delivery systems, lipid nanoparticles (LNPs) represent a remarkable innovation in cancer therapy. LNPs are lipid-based nano-delivery systems that can transport drugs or genetic material into the body (Alfutaimani et al., 2024). Such technology provides great control over drug release and precise delivery to intended sites. This stems from the phospholipid bilayer structure of these nanoparticles, which enables them to encapsulate drugs within the lipid bilayers or aqueous core (Alfutaimani et al., 2024). In addition to enabling targeted delivery, these nanoparticles are suitable for pharmaceutical manufacturing due to their scalability in production and easy sterilization (Alfutaimani et al., 2024).
Lipid nanoparticles appear in various structures and forms, depending on the choice of lipid types and synthesis techniques. One of these types is solid lipid nanoparticles (SLNs) which are made of lipids that are solid at room temperature. SLNs display multiple structural models depending on the distribution of drugs, such as drug-enriched shell, drug-enriched core, homogeneous matrix systems, and others (Alfutaimani et al., 2024). In addition to that, lipid nanoparticles can be present as nanostructured lipid carriers (NLCs), which have revealed better drug-loading efficiency while having a less ordered internal structure. These NLCs were created by combining both solid and liquid lipids in order to resolve issues related to lipid crystallinity and drug expulsion observed in SLNs (Alfutaimani et al., 2024). Moreover, another form of LNPs is lipid-drug conjugates (LDCs), which improve stability and controlled release profiles, and depend on the chemical bonding of therapeutic agents to lipid elements (Alfutaimani et al., 2024). Additionally, there are also polymer-lipid hybrid nanoparticles that combine the structural stability of polymers with the biocompatibility of lipids (Alfutaimani et al., 2024). This helps LNPs achieve increased adaptability. Hence, these diverse arrangements differently influence the stability, drug-loading abilities, and treatment efficacy of LNPs.
Download the full document
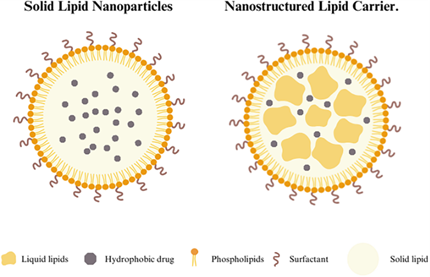
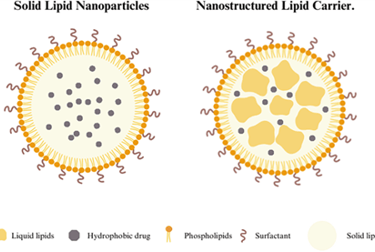
Figure 1. A simple visual aid illustrating Solid Lipid Nanoparticles (SLN) and Nanostructured Lipid Carriers (NLC) structures (Alfutaimani et al., 2024).
Mechanism Of Action
Understanding the mechanisms of action of such systems allows us to recognize their importance in targeted cancer treatment. One of the primary mechanisms of LNPs is passive transport through the enhanced permeability and retention (EPR) effect (Grigoletto et al., 2017). This method exploits the abnormal, permeable vasculature and deficient lymphatic drainage, which allows drug accumulation specifically at the tumor site. During this process, PEGylation, the attachment of polyethylene glycol (PEG) to drugs, improves their therapeutic efficacy by increasing their duration in the body, reducing immune reactions, and increasing their stability (Grigoletto et al., 2017).
Another mechanism that achieves further precision is active targeting which incorporates antibodies, peptides, or ligands designed to bind to tumor-specific receptors (Grigoletto et al., 2017). Upon localization at the tumor region, cellular uptake of LNPs takes place through endocytosis or membrane fusion. The internalization of the nanoparticles into tumor cells is facilitated by their small size and phospholipid structure (Grigoletto et al., 2017). In addition to the above-mentioned methods of transport, drug release can be highly controlled and restricted only to certain conditions associated with the tumor.
The responsiveness of the LNPs can be tailored to respond to various stimuli, including pH, temperature, and enzymatic activity that trigger lipid degradation (Grigoletto et al., 2017).
Lipid Nanoparticles in Cancer
While lipid nanoparticles are highly efficient drug transport systems, their function gains further significance when tackling the most widespread and hard-to-treat cancers.
Lung Cancer
To this day, lung cancer remains one of the most prevalent types of cancer, associated with high mortality rates. Despite the significant advances in chemotherapy, immunotherapy, and radiotherapy, lung cancer treatment is limited by various issues, including cytotoxicity, lack of specificity, and drug resistance (Omidian et al., 2024). Hence, this highlights the role of LNPs in addressing the need for new delivery methods. Lipid nanoparticles have gained increased interest for lung cancer therapies. This is because of their biocompatible, biodegradable, low-toxicity, and non-irritating properties. They help in encapsulation and controlled release of various therapeutic agents, which improves bioavailability. They also assist in drug transport through the bloodstream and across biological barriers (Omidian et al., 2024). In addition, inhalable nanoparticles enhance drug delivery by protecting drugs from degradation, enabling DNA/RNA delivery, prolonging circulation time, and supporting targeted delivery and imaging applications, thereby increasing treatment precision in lung cancer (Sakkal et al., 2025).
Hence, lipid nanoparticle formulations of widely used chemotherapeutic drugs, including paclitaxel, docetaxel, and cisplatin, have shown significant improvements in clinical outcomes. For example, paclitaxel-loaded lipid nanocarriers revealed enhanced cytotoxicity and therapeutic effectiveness against lung cancer in several studies (Omidian et al., 2024). To add, PLGA-lipid hybrid nanoparticles successfully provided a strategy to overcome paclitaxel resistance in lung cancer cells and showcased strong antitumor effects both in vitro and in vivo (Omidian et al., 2024). As shown in Figure 2, this approach allows the targeting of circulating anoikis (apoptosis)-resistant tumor cells by improving paclitaxel delivery to these drug-resistant cells. Moreover, RGD peptide-modified redox-sensitive lipid-polymer nanoparticles enhanced antitumor activity and further mitigated systemic toxicity, as RGD targets integrin receptors, especially 𝛂v𝛃3 and 𝛂v𝛃5 integrins that are overexpressed on tumor cells and blood vessels (Omidian et al., 2024). This demonstrates the potential of these platforms for targeting resistant lung cancer.
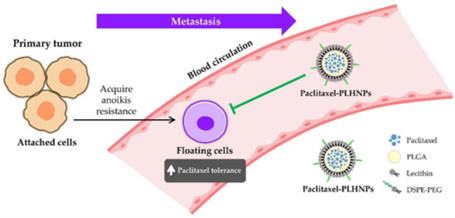
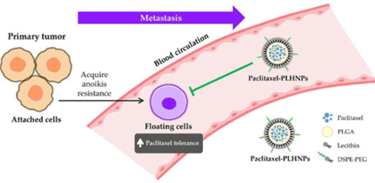
Figure 2. Paclitaxel-loaded PLGA–lipid hybrid nanoparticles (PLHNPs) help deliver paclitaxel to anoikis‑resistant circulating lung cancer cells during metastasis (Omidian et al., 2024).
Brain Cancer
Brain tumors are a result of the uncontrolled growth of neural or glial cells. They are especially hard to diagnose and manage due to the absence of early-stage symptoms, which delays detection and allows for further progression and worsening of the disease. Chemotherapy, one of the most commonly used treatments, has several limitations. One of the major restrictions is the blood-brain barrier (BBB), as it limits the delivery of both therapeutic and imaging agents to the tumor, leading to low drug levels and reduced effectiveness in treating this type of cancer (Mohanta et al., 2019). Lipid nanoparticles serve as an effective solution to these limitations. These nanoparticles allow for improved drug transport across the blood-brain barrier and decreased reticuloendothelial system clearance through surface modifications (Mohanta et al., 2019). Thus, they increase the accumulation of drugs in brain regions and decrease treatment doses, lowering the risk of adverse side effects.
Lipid-based enhancements further improve drug delivery across the BBB without damaging its integrity. Lipid‑vector prodrug methods, which involve attaching active drugs to lipid carriers, allow for targeted delivery of neuropeptides and other molecules straight to the brain (Mohanta et al., 2019). Furthermore, liposomal encapsulation of peptides and nanoparticles, especially charged ones, has shown a significant increase in delivery efficiency to the brain. Notably, as seen in the study by Mohanta et al. (2019), in vivo imaging studies have confirmed that advanced lipid nanoparticle systems have demonstrated efficient BBB passage and central nervous system uptake.
Moreover, further brain specificity is achieved by attaching targeting ligands to PEG chains or nanoparticle surfaces, allowing for specific receptor binding (Mohanta et al., 2019). Hence, these characteristics make lipid nanoparticles a highly effective approach to overcome the limitations posed by conventional therapies in the treatment of brain tumors.
Limitations of Lipid Nanoparticles
Lipid nanoparticles have various issues that limit their application in medicine. Because their manufacturing necessitates precise control over particle size, surface characteristics, and drug release behaviors, it is challenging to produce them on a large scale and ensure that each batch is identical (Omidian et al., 2024). In addition to that, concerns regarding immunological reactions persist despite continuous advancements. This implies the need for extensive safety testing for diverse patient populations (Omidian et al., 2024).
Clinical application is also hindered due to the strict regulatory requirements for the unique composition of LNPs which are often costly and time-consuming to meet. Even though preclinical studies often show promising results, clinical outcomes may not always align with expectations and can differ greatly from patient to patient due to biological complexity and patient-specific factors (Omidian et al., 2024). In addition, stability issues, such as sensitivity to storage conditions and lipid oxidation, represent significant limitations that can affect the validity and reliability of these formulations (Nomani et al., 2026b). Furthermore, although they appear to be beneficial at first, PEGylated nanoparticles can sometimes trigger an “accelerated blood clearance” (ABC) effect after repeated doses, as the immune system may recognize PEG as foreign and produce antibodies, reducing their effectiveness over time (Lila et al., 2013).
Notably, the blood–brain barrier penetration remains a significant clinical challenge; although lipid nanoparticles can improve drug delivery across the BBB, they do not overcome this barrier completely, restricting therapeutic efficacy in cases of brain metastases. Thus, these factors limit the predictability and generalizability of such advanced treatments.
Gold nanoparticles
Gold nanoparticles (AuNPs) have attracted researchers and physicians because they are easily synthesized and controlled. They have properties derived from gold, one of the least reactive chemical elements, and optimized methods of synthesis that enable the control of their shape, size, and dimensions (Sztandera et al., 2018). These nanoparticles exhibit different properties than their larger counterparts. For example, colloidal gold is more reactive than bulk gold, which enables it to have diverse interactions within biological systems and be used for various applications (Sztandera et al., 2018). Most importantly, AuNPs have surface plasmon resonance (SPR), a phenomenon that occurs when the oscillation of free electrons on the surface of the nanoparticle resonates with the frequency of incoming light radiation, creating a plasmon band (Sztandera et al., 2018). This phenomenon allows for surface-enhanced optical properties and enables the use of AuNPs in photothermal therapy.
There are different forms of AuNPs. To begin with, nanospheres are the most used type in biomedicine because of their relatively easy synthesis and their sizes ranging from 1 nm to 100 nm (Kalashgrani & Javanmardi, 2022). They also have strong visible light absorbance, which limits them to applications for superficial cancers as visible light does not reach deep body layers (Kalashgrani & Javanmardi, 2022). Another type of AuNPs is nanocages. These nanocages are hollow structures with porous walls that are formed from the galvanic substitution between nanocubes and gold chloride salt (Kalashgrani & Javanmardi, 2022). The third type is nanorods. These structures have controllable absorption in the near-infrared region and two absorption peaks because of the presence of two SPRs, one across and one along the nanorod. These optical properties enable the nanorod to be used in photothermal therapy and allow it to be tailored to the specific need for medical applications.
Mechanism Of Action
To efficiently and effectively integrate AuNPs in cancer treatments, we need to understand how they behave in the body. Spherical AuNPs were shown to generally localize in the liver, spleen, kidney, and lungs with some variation depending on size and shape (Sztandera et al., 2018). Importantly, these particles have been shown to penetrate the blood-brain barrier (BBB) by various mechanisms, such as endocytosis, passive diffusion, passage through ion channels, or disruption of tight junctions (Sztandera et al., 2018). Studies have also shown that particles ranging from 1.4 to 18 nm in size can pass across the placenta barrier and reach the fetus, through hypothesized mechanisms involving transcellular processes or transtrophoblastic channels (Sztandera et al., 2018).
Moreover, surface modifications determine the biological behavior of AuNPs; they help evade immune recognition and allow the particles to reach their site of action. Such functionalization can be achieved by the covalent attachment of ligands to the nanoparticle surface. For example, poly(ethylene glycol) (PEG) is widely used because of improved circulation time, reduced toxicity, and decreased nonspecific organ accumulation (Sztandera et al., 2018). Other ligands, like folic acid, can also be used to enhance biodistribution, targeted delivery, and cellular internalization (Sztandera et al., 2018).
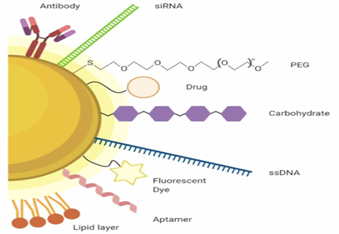
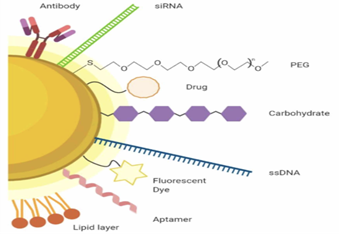
Figure 3. Gold nanomaterial functionalization strategies and coatings (Guinart et al., 2020)
Gold Nanotechnology In Cancer
The above mechanisms of action can be further enhanced and used in the treatment and diagnosis of different types of cancers.
Lung Cancer
As mentioned previously, lung cancer is one of the most fatal malignancies, and conventional therapies lack precision and can lead to significant systemic toxicity. These limitations have increased interest in the use of AuNPs in the treatment of lung cancer. As explained before, AuNPs can ensure the delivery of drugs to the target site. This can be used in the treatment of non-small cell lung cancer (NSCLC) that arises from mutations in the tyrosine kinase domain of the EGFR gene. Tyrosine kinase inhibitors such as afatinib are not used extensively because they have low tumor accumulation and systemic side effects (Guinart et al., 2020). However, Cryer et al. (2019) designed an Afatinib-GNP system that showed a 3.7-fold increase in potency when administered to lung cancer cells. They also examined the cytotoxicity of Afb-A and Afb-AuNPs on normal cells such as alveolar epithelial cells and found that Afb-A showed significant cytotoxicity while Afb-AuNPs showed minimal cytotoxicity (Cryer et al., 2019).
Another application of gold nanoparticles is through the attachments of SiRNAs. SiRNAs can be attached to AuNPs, thus increasing their circulation time and preventing their degradation by RNases (Guinart et al., 2020). For example, when AuNPs conjugated with c-Myc siRNA were introduced to a murine lung carcinoma xenograft model, they suppressed tumor growth (Kim et al., 2017). Such examples illustrate the potential of AuNPs in enhancing the treatment of lung cancer.
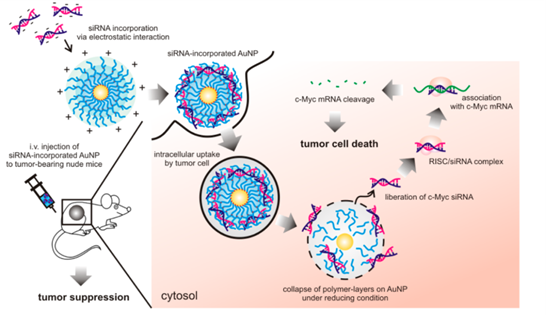
Figure 4. Schematic illustration of proposed anti-cancer effect of c-Myc siRNA-incorporated AuNP and cellular uptake process in human tumor xenograft mouse (Kim et al., 2017).
Brain Cancer
Brain cancer, specifically glioblastoma multiform (GBM), is one of the most invasive and hard-to-treat cancers because of the presence of the BBB and its high rate of recurrence. As a result, conventional treatments often fail to contain and prevent tumor recurrence. This has necessitated the search for different approaches, among which AuNPs have emerged as a promising alternative. Kumthekar et al. (2021) conducted a Phase 0, first-in-human trial on patients with recurrent GBM to test the safety and efficacy of NU-0129, a gold nanoparticle–based RNA interference therapeutic. The study revealed that the drug did not lead to significant toxicity in patients, downregulated GBM oncogene expression, and increased wild-type p53. Another study tested AuNPs conjugated with PEG, folate, anti-transferrin antibody, and curcin. Curcin is a type I ribosome-inactivating protein that can target glioma cells (Mohamed et al., 2013). The study revealed the ability of the drug to be selectively released in an acidic pH that mimics the tumor microenvironment and to kill 97% of glioma cells in-vitro after 48 h (at 100µg/ml). This shows the high efficiency and selectivity of such nanoparticles. Moreover, AuNPs can be used with radiotherapy to enhance the efficacy of the treatment. AuNPs can “increase X-ray absorption and generate local hyper heat for the thermal ablation of tumors” (Tu et al., 2021). This amplifies the radiotherapy effect and leads to pro-apoptotic unfolded protein responses (Tu et al., 2021). Tu et al. (2021) also showed that this combination can overcome tumor resistance, as no tumor recurrence was observed in a glioma xenograft nude mouse model after 14 days of treatment. Although these examples may not be clinically approved yet, they highlight the potential of AuNPs as both a combination and targeted therapy.
Limitations of Gold Nanoparticles
Gold nanoparticles have strong potential for use in cancer treatment; however, it is important to discuss the challenges faced when using such technology. Depending on the size, structure, and surface chemistry, AuNPs can lead to different toxicities, which are still not adequately characterized because of the lack of standardized assays for toxicity assessment (Singh et al., 2018). Moreover, the accumulation of AuNPs in organs presents a major concern. For example, Li et al. (2018) report that PEG-coated AuNPs accumulate in the spleen and liver. They also have a long retention time, resulting in a slow elimination rate (Li et al., 2018). This, in the long term, can lead to safety concerns and increased organ burden. Finally, the high cost of precious metals represents an additional barrier to the clinical application and large-scale production of such therapies.
Conclusion
In conclusion, nanotechnology is a wide field that has significant potential in the treatment of cancer. Unlike conventional therapies, it helps deliver medications to targeted tissues, thereby decreasing systemic toxicity. Moreover, the ability to control nanoparticle size, shape, and attach molecules to their surfaces makes them versatile and applicable across a wide range of cancer types. This is particularly important in the context of lung and brain cancers, as conventional therapies are hindered by severe side effects, lack of precision, and drug resistance. As presented above, different nanoparticles have different characteristics and mechanisms of action.
In particular, AuNPs have shown potential in the treatment of lung and brain cancer through targeted delivery of therapeutic agents, gene silencing, and, in some cases, combination with radiotherapy. Furthermore, lipid nanoparticles also demonstrate significant advancements in these cancers, as they enable targeted release of therapeutic agents, enhance cellular uptake, and reduce systemic toxicity. Most importantly, in the treatment of brain cancer, both lipid and gold nanoparticles have an advantage over some chemotherapies, as they can cross the blood-brain barrier more effectively.
Despite these advantages, nanoparticle production is expensive and time-consuming. It also requires a long process of preclinical and clinical testing, which does not always guarantee positive results. This may reduce industrial investment and hinder the progress of nanoparticle-based therapies. In addition, there is still insufficient data on the long-term effects of nanoparticles on patients, and further research into the effectiveness of nanoparticles on heterogeneous tumors is needed.
References
Alfutaimani, A. S., Alharbi, N. K., Alahmari, A. S., Alqabbani, A. A., & Aldayel, A. M. (2024). Exploring the landscape of Lipid Nanoparticles (LNPs): A comprehensive review of LNPs types and biological sources of lipids. International Journal of Pharmaceutics X, 8, 100305. https://doi.org/10.1016/j.ijpx.2024.100305
Cryer, A. M., Chan, C., Eftychidou, A., Maksoudian, C., Mahesh, M., Tetley, T. D., Spivey, A. C., & Thorley, A. J. (2019). Tyrosine kinase Inhibitor Gold nanoconjugates for the treatment of Non-Small Cell Lung Cancer. ACS Applied Materials & Interfaces, 11(18), 16336–16346. https://doi.org/10.1021/acsami.9b02986
Grigoletto, A., Mero, A., Maso, K., & Pasut, G. (2017). Transglutaminase-Mediated nanoarmoring of enzymes by PEGylation. Methods in Enzymology on CD-ROM/Methods in Enzymology, 590, 317-346. https://doi.org/10.1016/bs.mie.2017.01.002
Guinart, A., Perry, H. L., Wilton-Ely, J. D. E. T., & Tetley, T. D. (2020). Gold nanomaterials in the management of lung cancer. Emerging Topics in Life Sciences, 4(6), 627–643. https://doi.org/10.1042/etls20200332
Hazarika, D., Sarma, S., & Shankarishan, P. (2024). Nanotechnology in cancer therapeutics, diagnosis,and management. BioTechnologia, 105(3), 287–303. https://doi.org/10.5114/bta.2024.141807
Kalashgrani, M.Y.; Javanmardi, N. Multifunctional Gold nanoparticle: As novel agents for cancer treatment. Adv. Appl. Nano Bio-Technol. 2022, 3, 1–6.
https://doi.org/10.47277/AANBT/3(3)6
Kim, H. S., Son, Y. J., Mao, W., Leong, K. W., & Yoo, H. S. (2017). Atom transfer radical polymerization of multishelled cationic corona for the systemic delivery of SIRNA. Nano Letters, 18(1), 314–325. https://doi.org/10.1021/acs.nanolett.7b04183
Kumthekar, P., Ko, C. H., Paunesku, T., Dixit, K., Sonabend, A. M., Bloch, O., Tate, M., Schwartz, M., Zuckerman, L., Lezon, R., Lukas, R. V., Jovanovic, B., McCortney, K., Colman, H., Chen, S., Lai, B., Antipova, O., Deng, J., Li, L., . . . Stegh, A. H. (2021). A first-in-human phase 0 clinical study of RNA interference–based spherical nucleic acids in patients with recurrent glioblastoma. Science Translational Medicine, 13(584). https://doi.org/10.1126/scitranslmed.abb3945
Li, X., Hu, Z., Ma, J., Wang, X., Zhang, Y., Wang, W., & Yuan, Z. (2018). The systematic evaluation of size-dependent toxicity and multi-time biodistribution of gold nanoparticles. Colloids and Surfaces B Biointerfaces, 167, 260–266. https://doi.org/10.1016/j.colsurfb.2018.04.005
Lila, A. S. A., Kiwada, H., & Ishida, T. (2013). The accelerated blood clearance (ABC) phenomenon: Clinical challenge and approaches to manage. Journal of Controlled Release, 172(1), 38–47. https://doi.org/10.1016/j.jconrel.2013.07.026
Misra, R., Acharya, S., & Sahoo, S. K. (2010). Cancer nanotechnology: application of nanotechnology in cancer therapy. Drug Discovery Today, 15(19–20), 842–850. https://doi.org/10.1016/j.drudis.2010.08.006
Mohamed, M. S., Veeranarayanan, S., Poulose, A. C., Nagaoka, Y., Minegishi, H., Yoshida, Y., Maekawa, T., & Kumar, D. S. (2013). Type 1 ribotoxin-curcin conjugated biogenic gold nanoparticles for a multimodal therapeutic approach towards brain cancer. Biochimica Et Biophysica Acta (BBA) - General Subjects, 1840(6), 1657–1669. https://doi.org/10.1016/j.bbagen.2013.12.020
Mohanta, B. C., Palei, N. N., Surendran, V., Dinda, S. C., Rajangam, J., Deb, J., & Sahoo, B. M. (2019). Lipid based nanoparticles: Current strategies for brain tumor targeting. Current Nanomaterials, 4(2), 84–100. https://doi.org/10.2174/2405461504666190510121911
Nomani, A., Saraswat, A., Brown, H., Kuo, J., Duong, H., Wu, J., Zhang, Y., Fu, Y., Moon, Y., Wahidi, S., Mejia, N., Hartford, S., Qiu, H., Rayaprolu, B., Bhalla, A., & Shameem, M. (2026b). Identifying key factors affecting mRNA-Lipid nanoparticles drug product formulation stability. Nanomaterials, 16(4), 268. https://doi.org/10.3390/nano16040268
Omidian, H., Gill, E. J., & Cubeddu, L. X. (2024). Lipid nanoparticles in lung cancer therapy. Pharmaceutics, 16(5), 644. https://doi.org/10.3390/pharmaceutics16050644
Sakkal, M., Abdelmoteleb, R. W. A., Ali, A. A., Jardan, Y. A. B., Löbenberg, R., & Sarfraz, M. (2025). Inhalable nanoparticle-based drug delivery system for non-small cell lung cancer therapy: promises and challenges. Saudi Pharmaceutical Journal, 33(6), 50. https://doi.org/10.1007/s44446-025-00046-y
Singh, P., Pandit, S., Mokkapati, V., Garg, A., Ravikumar, V., & Mijakovic, I. (2018). Gold nanoparticles in diagnostics and therapeutics for human cancer. International Journal of Molecular Sciences, 19(7), 1979. https://doi.org/10.3390/ijms19071979
Sztandera, K., Gorzkiewicz, M., & Klajnert-Maculewicz, B. (2018). Gold nanoparticles in cancer treatment. Molecular Pharmaceutics, 16(1), 1–23. https://doi.org/10.1021/acs.molpharmaceut.8b00810
Tu, L., Luo, Z., Wu, Y., Huo, S., & Liang, X. (2021). Gold-based nanomaterials for the treatment of brain cancer. Cancer Biology and Medicine, 18(2), 372–387. https://doi.org/10.20892/j.issn.2095-3941.2020.0524
World Health Organization: WHO. (2026, April 16). Cancer. https://www.who.int/news-room/fact-sheets/detail/cancer
